
The top three Prescribed Perspectives articles downloaded by health systems in June.
Is your health system DSCSA-ready?
Date
July 26, 2023
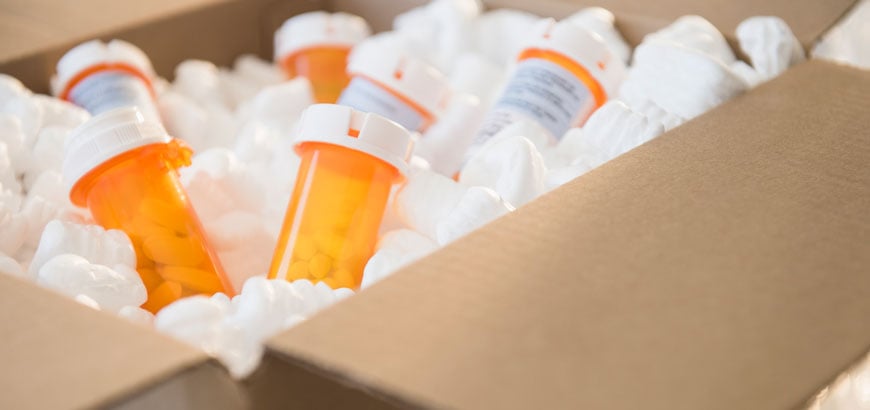
The Drug Supply Chain Security Act (DSCSA) is a US federal law that establishes requirements for the secure and traceable distribution of prescription drugs. Signed into law on November 27, 2013, it aims to enhance patient safety by preventing counterfeit, adulterated, or stolen drugs from entering the supply chain.
Is your health system DSCSA ready? Read the key compliance requirements or contact us to learn how McKesson can help you achieve more.


This visual helps illustrate the nuances of each type of bagging in specialty pharmacy and what it may mean for your health system.

As strategic partners, our Specialty Pharmacy Enablement experts can help drive improvements across the board.

In a unique, retreat-style setting, health systems leaders had close conversations around best practices, wins, struggles, and solutions.

Discover three key takeaways from a recent C-suite roundtable discussing the benefits and challenges of central fill for health systems.

Industry event highlights how connection, creativity, and a fresh perspective are shaping the future of health system innovation.

Our comprehensive and customizable solutions are deliberately designed with real customer challenges in mind.

McKesson’s Prateek Bhatia, VP/GM, Intrafusion, shares insights on the many opportunities health systems have with their infusion service line.

Expert insights on the rapid growth of specialty pharmacy and the importance of strategic integration.

![System.Func`1[System.String]](/siteassets/images/z-mckesson-logosicons/health-systems2.png)